Learning GOals
Acids & Bases Notes
Worksheets & Answer Keys
Study Cards
Review Packages
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Pages from Data Booklet
|
|
|
VIDEOS
|
|
|
|
Today in class...
Lesson 17: Ka Calculations
Prescribed Learning Outcomes: E3, E4 Study Guide Equivalent: M1 - M5
Today you reviewed the difference between strong and weak acids, writing Ka and Kb expressions, and using ICE tables to find the pH of a weak acid solution.
Homework:
- Finish Worksheet Problem Set #8: pH calculations for Weak Acids
- Hebden IV.14 # 74 - 83 pages 148 - 152
Prescribed Learning Outcomes: E3, E4 Study Guide Equivalent: M1 - M5
Today you reviewed the difference between strong and weak acids, writing Ka and Kb expressions, and using ICE tables to find the pH of a weak acid solution.
Homework:
- Finish Worksheet Problem Set #8: pH calculations for Weak Acids
- Hebden IV.14 # 74 - 83 pages 148 - 152
Lesson 16: Review of Mid-Unit Test
April 29, 30
Today, together as a class we reviewed the test, going over each question.
April 29, 30
Today, together as a class we reviewed the test, going over each question.
Lesson 15: Mid-Unit Test
April 24, 28
Today you wrote the Acid & Base mid unit test that covered sections IV.1 - IV.13.
April 24, 28
Today you wrote the Acid & Base mid unit test that covered sections IV.1 - IV.13.
Lesson 14: Review Class
Prescribed Learning Outcomes: Study Guide Equivalent:
Today you worked with a partner through a series of sample questions as preparation for the upcoming test.
Homework:
- STUDY!!!!! Please arrive to next class on-time and well prepared for the test
Prescribed Learning Outcomes: Study Guide Equivalent:
Today you worked with a partner through a series of sample questions as preparation for the upcoming test.
Homework:
- STUDY!!!!! Please arrive to next class on-time and well prepared for the test
Lesson 13: Hydrolysis Lab - 20D
April 20, 21
Prescribed Learning Outcomes: F4, F5 Study Guide Equivalent: N1 - N4
Homework:
- Hebden IV.13 p. 148 # 69 - 73
- Review for Mid-Unit Test
April 20, 21
Prescribed Learning Outcomes: F4, F5 Study Guide Equivalent: N1 - N4
Homework:
- Hebden IV.13 p. 148 # 69 - 73
- Review for Mid-Unit Test
Lesson 12: Hydrolysis
Prescribed Learning Outcomes: F4, F5 Study Guide Equivalent: N1 - N4
Today you had the opportunity to work together through a series of hydrolysis questions. The last 10 minutes in class were spent going over the answers. I also passed out the mid-unit review package.
Homework:
- Hebden IV.13 p. 148 # 69 - 73
- Review for Mid-Unit Test
Prescribed Learning Outcomes: F4, F5 Study Guide Equivalent: N1 - N4
Today you had the opportunity to work together through a series of hydrolysis questions. The last 10 minutes in class were spent going over the answers. I also passed out the mid-unit review package.
Homework:
- Hebden IV.13 p. 148 # 69 - 73
- Review for Mid-Unit Test
Lesson 11: Hydrolysis
Prescribed Learning Outcomes: F4, F5 Study Guide Equivalent: N1 - N4
Today you wrote a quiz involving pH and pOH calculations. After the quiz you saw that hydrolysis of a salt is a reaction between water and the cation or anion (or both) contained in the salt so as to produce an acidic or basic solution. The procedure for determining the behaviour of a salt in water is straightforward... a) determine ions produced when the salt dissociates b) discard spectator ions from further consideration c) any remaining ions will act as acids if they are on the acid side (left) of the table and/or as a base if they are on the base side (right). Here's a link to some online examples and a quiz for you to try. Want additional notes?
Homework:
- Hebden IV.13 p. 148 # 69 - 73
Prescribed Learning Outcomes: F4, F5 Study Guide Equivalent: N1 - N4
Today you wrote a quiz involving pH and pOH calculations. After the quiz you saw that hydrolysis of a salt is a reaction between water and the cation or anion (or both) contained in the salt so as to produce an acidic or basic solution. The procedure for determining the behaviour of a salt in water is straightforward... a) determine ions produced when the salt dissociates b) discard spectator ions from further consideration c) any remaining ions will act as acids if they are on the acid side (left) of the table and/or as a base if they are on the base side (right). Here's a link to some online examples and a quiz for you to try. Want additional notes?
Homework:
- Hebden IV.13 p. 148 # 69 - 73
Lesson 10: Advanced pH and pOH calculations
Prescribed Learning Outcomes: E2 Study Guide Equivalent: L8 - L12
Today you saw that mixing an acid and a base produces a solution which can be acidic, basic or neutral depending on the relative amounts of reactants involved. By determining the moles of acid or base in excess will then allow you to determine the pH of the solution. Want additional notes?
Homework:
- Hebden IV.12 p. 143 - 144 # 58 - 68
Prescribed Learning Outcomes: E2 Study Guide Equivalent: L8 - L12
Today you saw that mixing an acid and a base produces a solution which can be acidic, basic or neutral depending on the relative amounts of reactants involved. By determining the moles of acid or base in excess will then allow you to determine the pH of the solution. Want additional notes?
Homework:
- Hebden IV.12 p. 143 - 144 # 58 - 68
Lesson 9: pH & pOH
Prescribed Learning Outcomes: E2 Study Guide Equivalent: L8 - L12
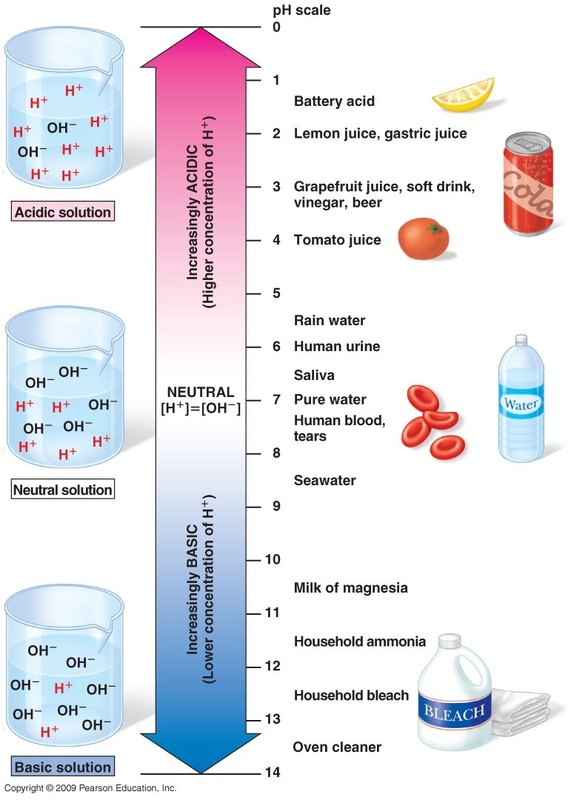
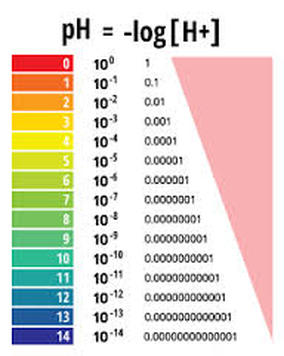
Today you learned that pH is a measure of the [H3O+] present in a solution. You also saw how the pH scale is a logarithmic scale used for convenience. You watched the pH and pOH: Crash Course Chemistry video. The last 20 minutes in class were spent completing the table for a variety of every day substances. Want additional notes?
Homework:
- Worksheet (7 questions) converting between pH, pOH, [H3O+], [OH-]
- Hebden IV.11 p. 134 - 141 # 47 - 57
Prescribed Learning Outcomes: E2 Study Guide Equivalent: L8 - L12
Today you learned that pH is a measure of the [H3O+] present in a solution. You also saw how the pH scale is a logarithmic scale used for convenience. You watched the pH and pOH: Crash Course Chemistry video. The last 20 minutes in class were spent completing the table for a variety of every day substances. Want additional notes?
Homework:
- Worksheet (7 questions) converting between pH, pOH, [H3O+], [OH-]
- Hebden IV.11 p. 134 - 141 # 47 - 57
Lesson 8: Ka & Kb
Prescribed Learning Outcomes: E3 Study Guide Equivalent: M1, M2
Today in class, you spent the first 20 minutes completing a review sheet for Kw. Next, you looked at both acid and base dissociation, how to write the reaction of a weak acid (or base) with water and how to write the ionization constant Ka (or Kb). The relationship between Ka and Kb was presented and you were shown the procedure for finding Kb. Want additional notes?
Homework:
- Hebden IV.8 p. 128 #31-34 & IV.9 p.130 #35-37
Prescribed Learning Outcomes: E3 Study Guide Equivalent: M1, M2
Today in class, you spent the first 20 minutes completing a review sheet for Kw. Next, you looked at both acid and base dissociation, how to write the reaction of a weak acid (or base) with water and how to write the ionization constant Ka (or Kb). The relationship between Ka and Kb was presented and you were shown the procedure for finding Kb. Want additional notes?
Homework:
- Hebden IV.8 p. 128 #31-34 & IV.9 p.130 #35-37
Lesson 7: Ion Product Constant of Water - Kw
Prescribed Learning Outcomes: E1 Study Guide Equivalent: L1 - L7
Today you handed in your lab write up for Experiment 20B. Next, you wrote a quiz on acids and bases. The lesson today showed how to write an equation to represent the ionization of water. Using the equation you were able to make predictions of how the addition of an acid or a base would affect the equilibrium system. You wrote the equilibrium expression for the ion product constant of water (Kw) and you examined how the value for Kw will change with changes in temperature. Finally you learned how to calculate the concentration of H3O+ (OH-) given the other, using Kw. Want additional notes?
Answers to multiple choice questions in notes: 4. D 5. A
Homework:
- Hebden IV.7 p.127 #29-30
Prescribed Learning Outcomes: E1 Study Guide Equivalent: L1 - L7
Today you handed in your lab write up for Experiment 20B. Next, you wrote a quiz on acids and bases. The lesson today showed how to write an equation to represent the ionization of water. Using the equation you were able to make predictions of how the addition of an acid or a base would affect the equilibrium system. You wrote the equilibrium expression for the ion product constant of water (Kw) and you examined how the value for Kw will change with changes in temperature. Finally you learned how to calculate the concentration of H3O+ (OH-) given the other, using Kw. Want additional notes?
Answers to multiple choice questions in notes: 4. D 5. A
Homework:
- Hebden IV.7 p.127 #29-30
Lesson 6: Bronsted-Lowry Acid and Base Equilibria
Prescribed Learning Outcomes: D5 Study Guide Equivalent: K6, K8, K9
Today you completed lab 20B where you observed the colour changes that occur with a number of different acid-base indicators and then you used the data to arrange all the B-L acids involved in the experiment in order of decreasing strength. Lab write up should include a data table, and answers to lab questions #1, 2 and follow-up questions 1, 2, and 3.
Homework:
- Finish lab write up
- Come to class ready for Quiz
Prescribed Learning Outcomes: D5 Study Guide Equivalent: K6, K8, K9
Today you completed lab 20B where you observed the colour changes that occur with a number of different acid-base indicators and then you used the data to arrange all the B-L acids involved in the experiment in order of decreasing strength. Lab write up should include a data table, and answers to lab questions #1, 2 and follow-up questions 1, 2, and 3.
Homework:
- Finish lab write up
- Come to class ready for Quiz
Lesson 5: Relative Strengths of Acids and Bases
Prescribed Learning Outcomes: D5 Study Guide Equivalent: K6, K8, K9
Today you used the table of relative strengths to predict whether reactants or products are favoured. After the lesson, you worked in teams of three to complete 5 stations. Remaining time was spent working on homework questions #38-46. Want additional notes?
Answer to multiple choice question: Iv.10 1D
Homework:
- Read and look over examples for Section IV.10 in Hebden p. 130-133
- Do Hebden Questions: # 38 - 46 p.133
- Prepare for Acid/Base Quiz
Prescribed Learning Outcomes: D5 Study Guide Equivalent: K6, K8, K9
Today you used the table of relative strengths to predict whether reactants or products are favoured. After the lesson, you worked in teams of three to complete 5 stations. Remaining time was spent working on homework questions #38-46. Want additional notes?
Answer to multiple choice question: Iv.10 1D
Homework:
- Read and look over examples for Section IV.10 in Hebden p. 130-133
- Do Hebden Questions: # 38 - 46 p.133
- Prepare for Acid/Base Quiz
Lesson 4: "STRONG & WEAK" Acids and Bases
Prescribed Learning Outcomes: D5 Study Guide Equivalent: K6
Today, you were introduced to page 6 of the data booklet "Relative Strengths of Bronsted-Lowry Acids and Bases". You were given time to read pages 121-125, take your own notes and complete questions 21 to 27. Looking for more notes on this topic? Have a look at this or maybe these will be helpful! Just before the end of class a take home quiz was handed out to be completed for next class.
Homework:
- Finish notes on strong and weak acids and bases (Section IV.5 in Hebden)
- Finish Hebden Questions: # 21 - 27, pages 121 - 126
- Take home quiz - 8 questions
Prescribed Learning Outcomes: D5 Study Guide Equivalent: K6
Today, you were introduced to page 6 of the data booklet "Relative Strengths of Bronsted-Lowry Acids and Bases". You were given time to read pages 121-125, take your own notes and complete questions 21 to 27. Looking for more notes on this topic? Have a look at this or maybe these will be helpful! Just before the end of class a take home quiz was handed out to be completed for next class.
Homework:
- Finish notes on strong and weak acids and bases (Section IV.5 in Hebden)
- Finish Hebden Questions: # 21 - 27, pages 121 - 126
- Take home quiz - 8 questions
Lesson 3: Review of Sections IV.1 - IV.4
Prescribed Learning Outcomes: D1 - D3 Study Guide Equivalent: J2 - J12
Today, you reviewed how to name acids and bases (notes). Here are a couple of memory aids for naming oxy-acids:
I "ATE" at the Cafeteria and got s"IC"k
R"ITE"-"OUS" Check these out...Righteous Brothers and a clip from Ferris Bueller's Day Off
Next, you worked in teams of three and completed questions at 4 stations.
Last few minutes of class I shared a Ted Talk that has nothing to do with Chemistry.
Homework:
- Finish Hebden Questions: # 1 - 19, pages 110 - 121
Lesson 2: The Nature of H+ & the Bronsted-Lowry Theory of Acids and Bases
Prescribed Learning Outcomes: D2, D3 Study Guide Equivalent: J6, J7, J9, J10, J11, J12
Today, you reviewed the true nature of a proton in water (notes, m.c. answer key: 1.A 2.B) . Next, you watched the Crash Course Chemistry video on acids and bases in solution. The class compared the Arrhenius definition of acids and bases to the more general Bronsted-Lowry definition. Next, you saw how to identify conjugate acid/base pairs in typical Bronsted-Lowry acid-base reactions. New vocabulary was introduced including, monoprotic, diprotic, amphiprotic, amphoteric, and conjugate pair
Want notes? Click here. The remainder of the class was spent working on problem set #2.
Answers to multiple choice questions- Iv.4: 1.A 2.D 3.C , Iv.5: 1.B 2.C 3.D 4.B
Homework:
- Finish problem set #2
- Do Hebden Questions: # 10 - 19, pages 115 - 121
Lesson 1: Introduction to Acids and Bases
Prescribed Learning Outcomes: D1, D2 Study Guide Equivalent: J2, J3, J4, J5
Today was an introduction to unit IV. The class watched a video and then you had a few minutes to brainstorm and write down everything you remember learning about acids and bases. After sharing what you wrote with a neighbour, you looked at a variety of household products and pictures, discussing how they relate to acids and bases (get PPT here) . Next, you were asked whether you agree or disagree with 3 statements. You were shown a demo using litmus and bromothymol blue indicators to test the pH of HCl, deionized water, NaOH, NaCl, Na2CO3, and NH3. You then worked in groups of three to sort unit vocabulary terms into categories. The remainder of the class was spent working on problem set #1. Want notes? Click here.
Answers to multiple choice questions- 1.A 2.A 3.C 4.B
Homework:
- Finish problem set #1
- Do Hebden Questions: # 1 - 9, pages 110 - 114
Prescribed Learning Outcomes: D1 - D3 Study Guide Equivalent: J2 - J12
Today, you reviewed how to name acids and bases (notes). Here are a couple of memory aids for naming oxy-acids:
I "ATE" at the Cafeteria and got s"IC"k
R"ITE"-"OUS" Check these out...Righteous Brothers and a clip from Ferris Bueller's Day Off
Next, you worked in teams of three and completed questions at 4 stations.
Last few minutes of class I shared a Ted Talk that has nothing to do with Chemistry.
Homework:
- Finish Hebden Questions: # 1 - 19, pages 110 - 121
Lesson 2: The Nature of H+ & the Bronsted-Lowry Theory of Acids and Bases
Prescribed Learning Outcomes: D2, D3 Study Guide Equivalent: J6, J7, J9, J10, J11, J12
Today, you reviewed the true nature of a proton in water (notes, m.c. answer key: 1.A 2.B) . Next, you watched the Crash Course Chemistry video on acids and bases in solution. The class compared the Arrhenius definition of acids and bases to the more general Bronsted-Lowry definition. Next, you saw how to identify conjugate acid/base pairs in typical Bronsted-Lowry acid-base reactions. New vocabulary was introduced including, monoprotic, diprotic, amphiprotic, amphoteric, and conjugate pair
Want notes? Click here. The remainder of the class was spent working on problem set #2.
Answers to multiple choice questions- Iv.4: 1.A 2.D 3.C , Iv.5: 1.B 2.C 3.D 4.B
Homework:
- Finish problem set #2
- Do Hebden Questions: # 10 - 19, pages 115 - 121
Lesson 1: Introduction to Acids and Bases
Prescribed Learning Outcomes: D1, D2 Study Guide Equivalent: J2, J3, J4, J5
Today was an introduction to unit IV. The class watched a video and then you had a few minutes to brainstorm and write down everything you remember learning about acids and bases. After sharing what you wrote with a neighbour, you looked at a variety of household products and pictures, discussing how they relate to acids and bases (get PPT here) . Next, you were asked whether you agree or disagree with 3 statements. You were shown a demo using litmus and bromothymol blue indicators to test the pH of HCl, deionized water, NaOH, NaCl, Na2CO3, and NH3. You then worked in groups of three to sort unit vocabulary terms into categories. The remainder of the class was spent working on problem set #1. Want notes? Click here.
Answers to multiple choice questions- 1.A 2.A 3.C 4.B
Homework:
- Finish problem set #1
- Do Hebden Questions: # 1 - 9, pages 110 - 114